EOF in the Slit Pore Geometry¶
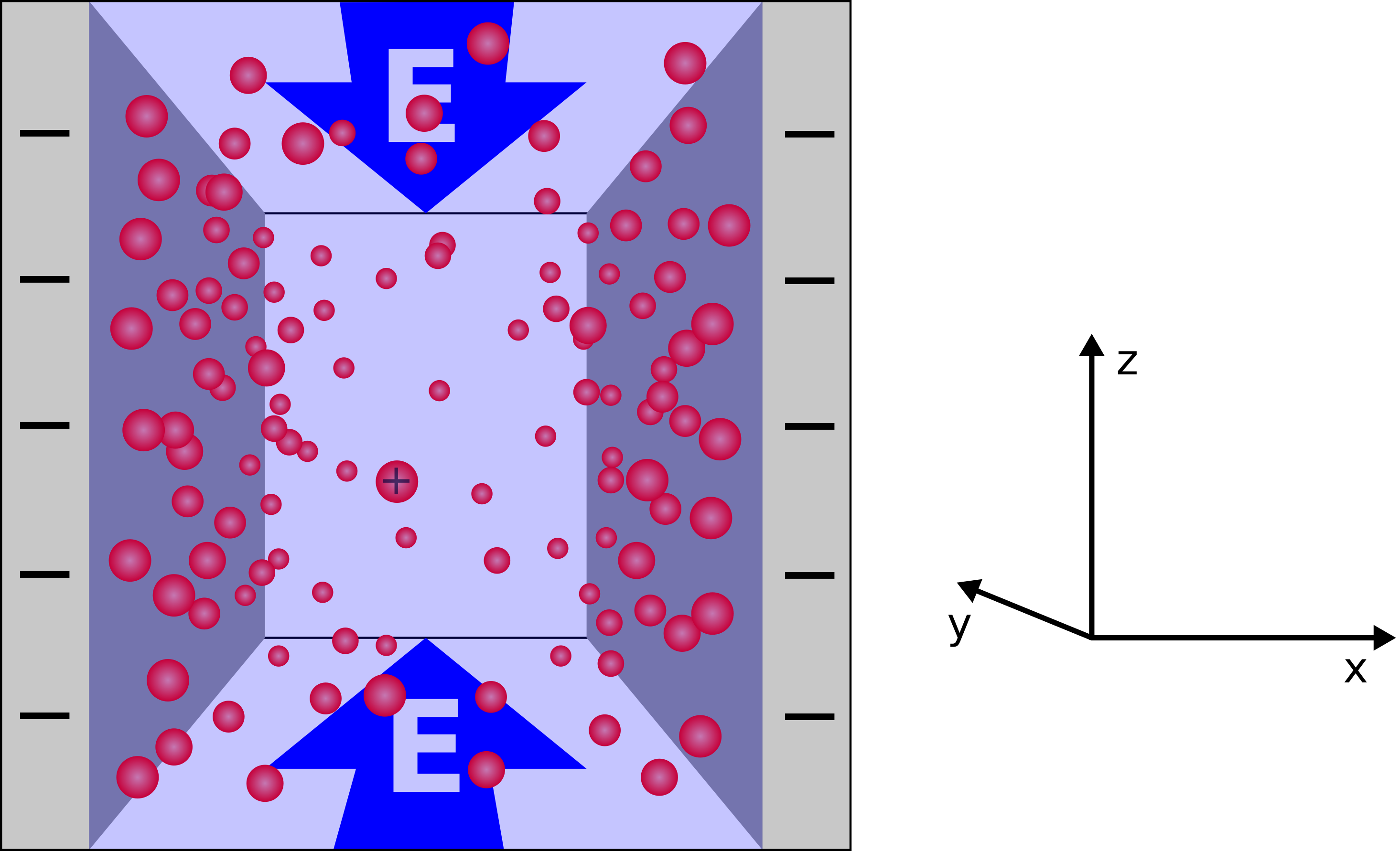
The slit pore system depicted in Fig. 1 consists of two like charged parallel plates of infinite extent, confining a solution of water and the plates' counterions.
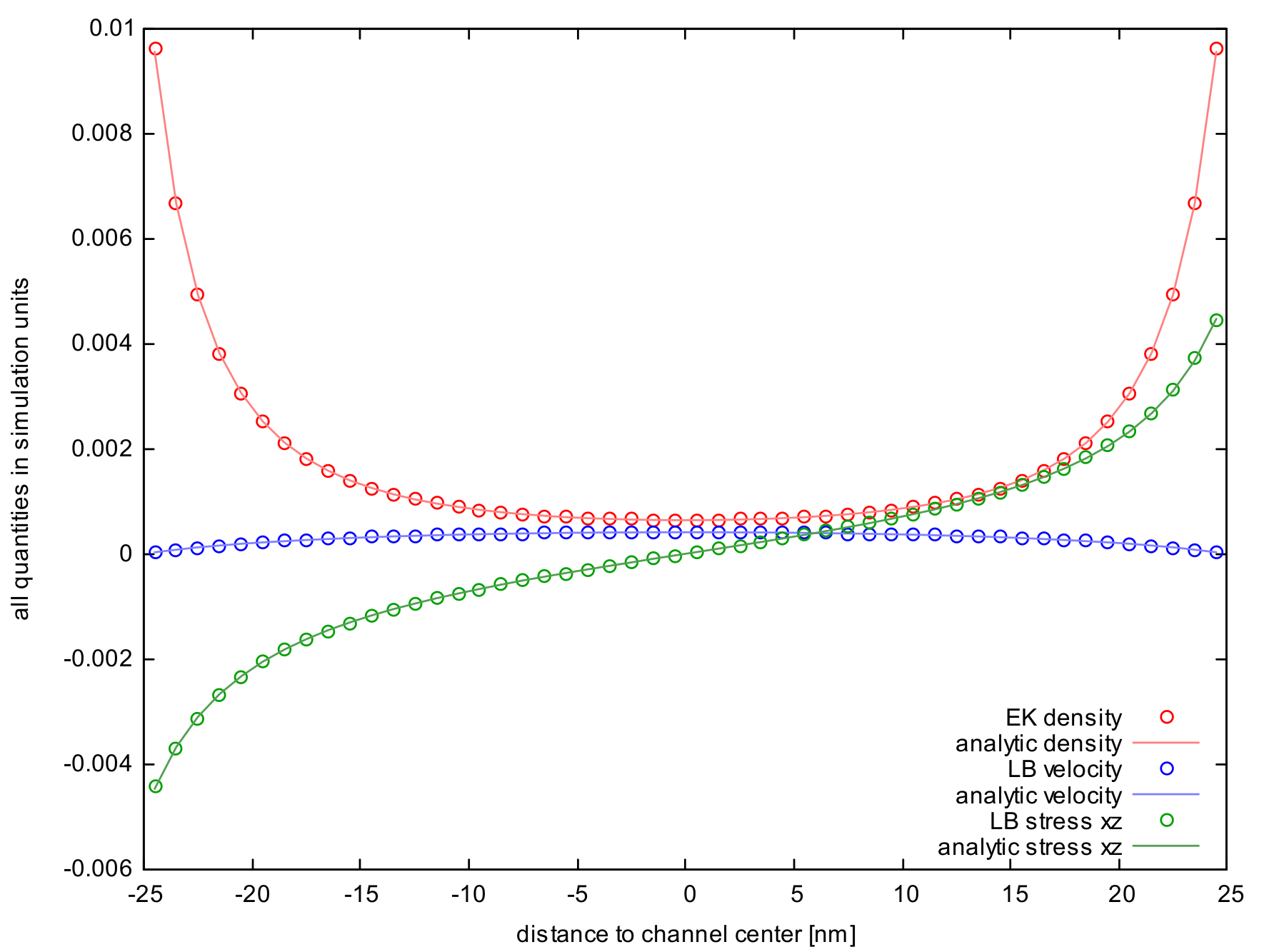
Due to the net neutrality of the system and the translational symmetry in directions parallel to the plates, the potential outside the two plates must be constant. This means that using periodic or non-periodic boundary conditions makes no difference. As the system is in equilibrium in the normal direction, the electrokinetic equations for this dimension reduce to the Poisson-Boltzmann equation for the electrostatic potential, which reads \begin{align} \partial_x^2 \Phi(x) = -4 \pi \, k_\mathrm{B}T \, l_\mathrm{B} \, ze \, c_0 \cdot \exp{\left(-\frac{ze\Phi(x)}{k_\mathrm{B}T}\right)} \; , \end{align} where $x$ denotes the direction normal to the plates. The constant $c_0$ has to be chosen such that charge neutrality is fulfilled. Multiplying by $2 \partial_x \Phi(x)$ and applying the inverse chain rule further reduces the equation to first order. Subsequent separation of variables yields the solution \begin{align} \Phi(x) = -\frac{k_\mathrm{B}T}{ze} \cdot \log \left[ \frac{C^2}{8 \pi \, k_\mathrm{B}T \, l_\mathrm{B}} \cdot \cos^{-2}\left( \frac{zeC}{2 k_\mathrm{B}T} \cdot x\right) \right], \quad \left| \frac{zeC}{2 k_\mathrm{B}T} \cdot x \right| < \frac \pi 2\; . \label{eq:validation_pb_counterions} \end{align} Refer to [5] for details on this calculation. Knowing that the counterion density $c$ resembles a Boltzmann distribution in the potential $ze \Phi$ leads to the expression \begin{align} c(x) = \frac{C^2}{8 \pi \, k_\mathrm{B}T \, l_\mathrm{B}} \cdot \cos^{-2} \left( \frac{zeC}{2 k_\mathrm{B}T} \cdot x \right) \; . \label{eq:validation_pb_density} \end{align} The constant $C$ is determined by fixing the number of counterions or requiring the E-field to vanish outside the volume contained by the plates. Both yields \begin{align} C \cdot \tan \left( \frac{zed}{4 k_\mathrm{B}T} \cdot C \right) = -4 \pi \, k_\mathrm{B}T \, l_\mathrm{B} \sigma \; , \end{align} where $d$ denotes the distance between the plates and $\sigma$ their (constant) surface charge density.
Applying an electric field along one of the directions parallel to the plates does not influence the charge distribution in the normal direction, which allows us to write down the hydrodynamic equations for the parallel direction. After eliminating all terms from the Navier-Stokes Equations, which vanish due to symmetry, we are left with \begin{align} \frac{\partial_x^2 v_y(x)}{\partial x^2} = -\frac{q E C^2}{8 \, k_\mathrm{B}T \, l_\mathrm{B} \, \eta} \cdot \cos^{-2} \left( \frac{qC}{2 k_\mathrm{B}T} \cdot x \right) \; , \end{align} which yields, by means of simple integration and the application of no-slip boundary conditions \begin{align} v_y(x) = \frac{E}{2 \pi \, l_\mathrm{B} \, \eta \, ze} \cdot \log \left[ \frac{\cos \left( \frac{zeC}{2 k_\mathrm{B}T} \cdot x \right)}{\cos \left( \frac{zeC}{2 k_\mathrm{B}T} \cdot \frac d 2 \right)} \right] \; . \end{align}
With this tutorial comes a Python script eof_analytical.py, which evaluates all these expressions on the same grid as is used in the upcoming simulation.